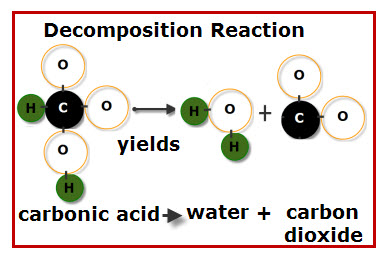
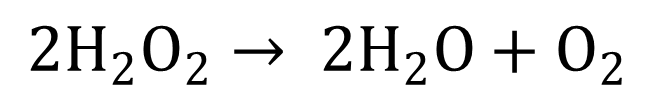- A decomposition reaction is a type of chemical reaction in which a single compound breaks down into two or more elements or new compounds. These reactions often involve an energy source such as heat, light, or electricity that breaks apart the bonds of compounds.
The chemical formula for decomposition is: AB----> A + B
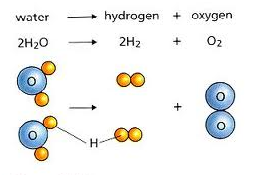
This is a decomposition reaction: 2 H2O ----> 2 H2 + O2
This is a decomposition reaction: 2 H2O ----> 2 H2 + O2
Characteristics of a decomposition reaction:
- one single reactant (compound) and two or more products
-requires energy
- one single reactant (compound) and two or more products
-requires energy
If you want to know more about decomposition you can go to these sites:
http://amrita.olabs.co.in/?sub=73&brch=3&sim=80&cnt=1
http://amrita.olabs.co.in/?sub=73&brch=3&sim=80&cnt=1